In This Article
- The Problem: Your Cells Have Power Plants That Can Break
- Why Doctors Couldn't Fix Them — Until Now
- How Does MitoCatch Actually Get Mitochondria Inside a Cell?
- What This Means for Patients With Mitochondrial Disease
- The Questions Still Unanswered
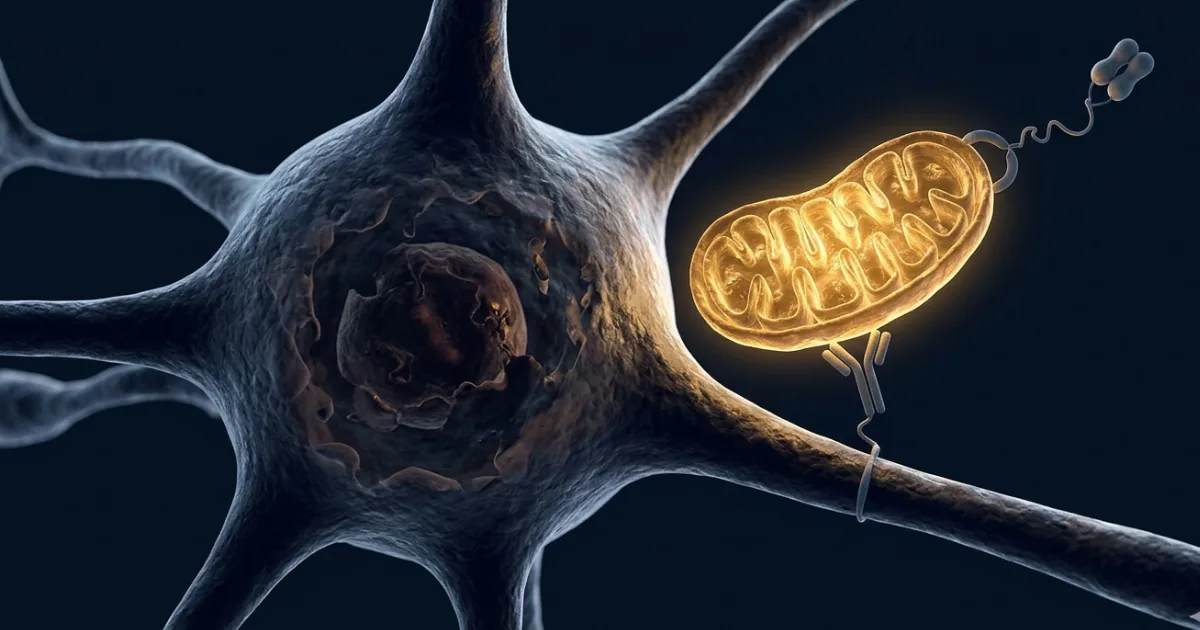
Imagine your body's cells slowly losing power — not because of a virus or a tumour, but because the tiny energy generators inside them are broken. That's what happens in dozens of diseases, from rare inherited blindness to heart failure. For decades, scientists knew the problem but had no clean way to fix it. Now a team at the Institute of Molecular and Clinical Ophthalmology Basel has published a study in Nature describing a system called MitoCatch — one that can guide healthy mitochondria into specific diseased cells, like a delivery drone with a built-in address label.
The Problem: Your Cells Have Power Plants That Can Break
Mitochondria are the structures inside your cells that convert food into usable energy. They're in almost every cell in your body — and in some tissues, like the heart and brain, they're absolutely critical. When they malfunction, the result can be devastating: nerve death, vision loss, muscle failure, and worse.
The tricky part is that mitochondrial diseases affect the organelle itself, which means you can't simply give someone a drug that repairs it. The damage is often encoded in mitochondrial DNA — a separate, ancient genetic system that has its own rules.
Why Doctors Couldn't Fix Them — Until Now
The idea of transplanting healthy mitochondria into damaged cells isn't new. Researchers have been trying it in labs for years, and there have even been some early human trials — mostly in heart surgery patients. But the results were always patchy. The mitochondria would enter cells somewhat randomly. There was no way to say: go to this cell type and not that one. In a complex organ like the retina, which has dozens of different cell types packed tightly together, that lack of precision was a serious problem.
Think of it like airdropping medicine over a city without any targeting system. Some reaches the right people. A lot doesn't.
How Does MitoCatch Actually Get Mitochondria Inside a Cell?
Here's the interesting part. Lead researcher Botond Roska and his colleagues didn't invent something entirely from scratch — they borrowed an idea from viruses. Some viruses enter specific cells because their surface proteins bind to matching proteins on the cell's outer membrane. MitoCatch works on the same principle, but the team engineered it in three different ways.
In one version (MitoCatch-C), they put a tiny "grabber" molecule on the surface of a target cell — a nanobody, essentially a miniature antibody — that latches onto proteins on the outside of donor mitochondria. In another version (MitoCatch-M), the grabber sits on the mitochondrion itself and seeks out a specific protein on the target cell. The third approach uses a bispecific binder: a molecule with one arm grabbing the mitochondrion and another grabbing the target cell, bridging the two.
In lab tests, 91% of neurons that displayed the right surface molecule ended up receiving donor mitochondria. That's not a small improvement over the old approach — it's a different category of result entirely.
"MitoCatch is a potential strategy to target disease-affected cell types with mitochondria in organs affected by diseases associated with mitochondrial dysfunction."
— Ayupov, Moreno-Juan et al., Institute of Molecular and Clinical Ophthalmology Basel · Nature, 2026What This Means for Patients With Mitochondrial Disease
The team tested MitoCatch on neurons derived from a real patient with LHON, a condition caused by a mutation in mitochondrial DNA that progressively kills the optic nerve. These are neurons that are already dying. After transplanting healthy mitochondria using MitoCatch, the treated neurons survived 23.6% better under stressful conditions compared to untreated ones. Their oxygen consumption — a direct measure of how much energy they're making — also went up significantly.
Then the team went further. They tested it in living mice. After deliberately crushing the optic nerve to simulate injury, mice whose retinal ganglion cells received targeted mitochondria showed 46.8% better cell survival after ten days compared to controls. Crucially, the surviving cells still responded to light — they were functioning, not just alive in name only.
The Questions Still Unanswered
The researchers are careful to say this is early-stage work. The mouse experiments showed cell survival at ten days — nobody yet knows if the benefit lasts weeks or months. The donor mitochondria used in these experiments came from lab-grown cells, and scaling that to clinical-grade production for human patients is a different challenge altogether.
There's also the question of immune reaction. Introducing foreign mitochondria into the body sounds like it should trigger inflammation, but the team's initial data — injecting mitochondria into mouse eyes, brains, and bloodstreams — showed no detectable antibody response. That's encouraging. But longer-term immune studies in humans will be essential before any clinical trial can begin.
What's clear is that the targeting problem — the one that made previous mitochondrial transplant attempts so imprecise — now has a credible solution. The next step, as the researchers note, is testing whether the protection holds over longer periods, and extending the approach to other organs beyond the eye.
- Targeted delivery works: MitoCatch can guide healthy mitochondria into specific cell types with over 90% efficiency — a massive improvement on untargeted approaches.
- Real patient cells responded: Neurons derived from an actual LHON patient survived significantly better after receiving donor mitochondria, with measurable gains in energy output.
- No immune alarm — so far: Animal studies showed no antibody response to transplanted mitochondria, which is promising for eventual human use, though much longer studies are needed.
"We have shown short- or mid-term neuroprotection by targeted mitochondrial transplantation in two cell types: LHON iHNeurons in vitro and mouse retinal ganglion cells in vivo." — Ayupov, Moreno-Juan et al., Nature, 2026.
📄 Source & Citation
Primary Source: Ayupov T, Moreno-Juan V, Curtoni S, et al. (2026). Cell-type-targeted mitochondrial transplantation rescues cell degeneration. Nature. https://doi.org/10.1038/s41586-026-10391-0
Authors & Affiliations: Temurkhan Ayupov & Verónica Moreno-Juan (co-first authors), Institute of Molecular and Clinical Ophthalmology Basel; with collaborators from ETH Zürich, University of Basel, Friedrich Miescher Institute, and University of Cambridge.
Data & Code: Proteomics dataset and scRNA-seq count matrix available via Zenodo: https://doi.org/10.5281/zenodo.17909630. Raw sequencing data via NCBI SRA under BioProject PRJNA1380367.
Key Themes: Mitochondrial Transplantation · Organelle Therapy · Nanobody Engineering · Leber Hereditary Optic Neuropathy · Retinal Neuroprotection
Supporting References:
[1] Borcherding N & Brestoff JR (2023). The power and potential of mitochondria transfer. Nature, 623, 283–291.
[2] Nakai R et al. (2024). Mitochondria transfer-based therapies reduce the morbidity and mortality of Leigh syndrome. Nat. Metab., 6, 1886–1896.
[3] Walker M et al. (2024). Mitochondrial transplantation in ischemic stroke: insights from a first-in-human brain trial. Curr. Transplant. Rep., 11, 53–62.
No comments yet. Be the first to share your thoughts.
Leave a Comment