In This Article
- The Molecular Error That Kills in Days
- Starting With the Cure, Not the Disease
- How Did Vitamin B3 Rescue a Dying Brain?
- What the Findings Mean for Real Patients
- The Questions That Still Need Answering
At birth, the mice looked normal. By day five, they were dying — stunted, brain-damaged, destined to be gone within the week. This is NAXD disease: a genetic disorder so catastrophic it kills human infants within months, and for which no approved treatment has ever existed. A team of researchers at the Gladstone Institutes just changed that calculus entirely — not with a designer drug, but with a vitamin you can buy at any pharmacy.
The Molecular Error That Kills in Days
Every cell in your body runs on a molecule called NADH — a kind of rechargeable battery that powers hundreds of metabolic reactions. But NADH is imperfect. Under the wrong temperature or pH, it spontaneously absorbs a water molecule and warps into a toxic form called NADHX. Think of it like a key that's been left out in the rain: it still fits in the lock, but now it jams the whole mechanism.
Your body has a dedicated repair enzyme — NAXD — whose only job is to catch these warped keys and straighten them back out. Lose NAXD, and the damaged molecules pile up. Toxic NADHX floods the cell. The molecular machinery chokes. And in the brain, where energy demand is relentless, cells start dying within days of birth.
Researchers have documented dozens of children with loss-of-function mutations in NAXD. Severe cerebral atrophy. Developmental failure. Death, typically within the first few months or years of life. Some children also develop skin lesions that look uncannily like pellagra — the disease caused by vitamin B3 deficiency. That skin clue was a breadcrumb nobody had followed. Until now.
Starting With the Cure, Not the Disease
The lab of Isha Jain at UC San Francisco made a deliberate choice to work backwards. Most medical research starts with a sick patient and asks: what drug fixes this? Jain's team started with a potential treatment — vitamins B2 and B3, individually — and asked: which genetic diseases would these vitamins rescue?
They ran genome-wide CRISPR screens: 200 million cells, each with a different gene knocked out, grown in media containing either normal or near-zero levels of one vitamin. After 15 days, they checked which knockouts died under low-vitamin conditions but survived under high-vitamin conditions. Cross-reference that list with every known human genetic disease, and you have a nomination list for vitamin-treatable disorders.
The vitamin B3 screen returned 15 disease candidates. At the very top sat NAXD. The connection made intuitive sense — vitamin B3 is the raw ingredient for making NAD, the molecule that NAXD is supposed to protect. If the protective enzyme is broken, flooding the system with more raw material might be enough to compensate. It was a hypothesis worth testing.
How Did Vitamin B3 Rescue a Dying Brain?
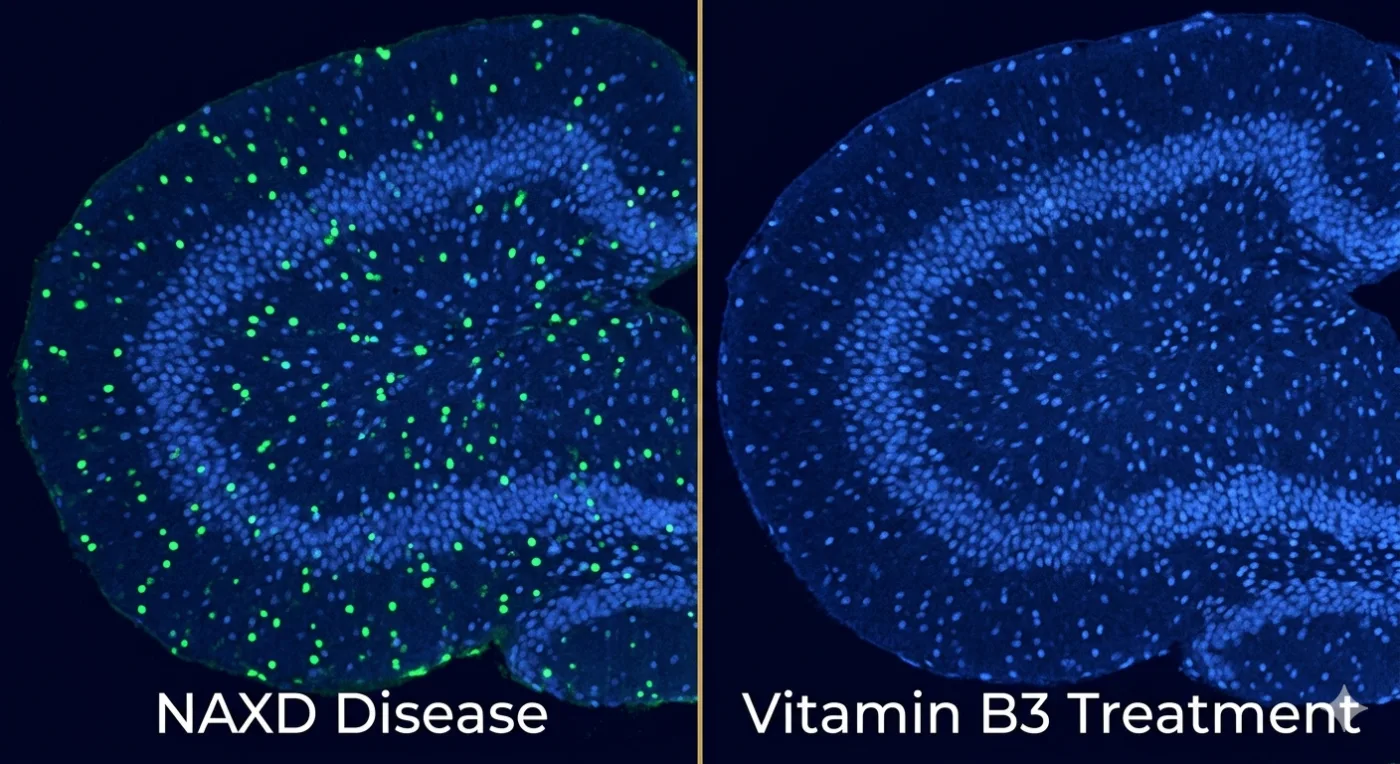
The researchers bred mice with frameshift mutations in the Naxd gene — knocking out every known isoform of the protein. Untreated knockout pups looked normal at birth, then collapsed. All were dead within seven days. Their brains showed the same cortical cell death and microglial inflammation seen in human NAXD patients by MRI. Their skin showed the same thinning seen in children with the disease. The model was accurate.
Then they tried vitamin B3 — specifically nicotinamide riboside, injected daily into the abdomen starting the moment pups were born. The result was not subtle. Eight of nine treated knockout mice survived past 200 days. They gained normal body weight. Their brains looked histologically healthy. Every measurable marker of NAXD disease — cell death, brain NAD depletion, toxic NADHX accumulation — reversed. The mice were, by every test the researchers ran, indistinguishable from their healthy littermates.
Metabolomics pointed to a specific downstream mechanism: serine. The brain uses an NAD-dependent enzyme called PHGDH to synthesize serine, an amino acid that feeds into dozens of other critical pathways. NAXD-deficient brains were dramatically depleted in serine. The toxic NADHX molecules directly inhibited PHGDH in biochemical assays — and vitamin B3 treatment restored serine levels alongside NAD. A single-nuclei RNA sequencing dataset of nearly 400,000 brain cells then identified the most vulnerable cell type: brain endothelial cells, the thin lining of cerebral blood vessels, showed the most dramatic transcriptional distress — and the most complete recovery after treatment.
"Rather than beginning with a disease and searching for a cure, we started with a potential cure and identified the diseases it could potentially treat."
— Garg, Blume, Jain et al., Gladstone Institutes · Cell, 2026What the Findings Mean for Real Patients
This research matters for several reasons that go well beyond a single rare disease. Vitamin B3 — whether as niacin, nicotinamide, or nicotinamide riboside — is cheap, widely available, and carries a well-established safety record. It already has regulatory approval for treating high cholesterol at doses of 1,000–3,000 mg per day in adults. The treatment here isn't some future drug waiting a decade for approval. It's on the shelf right now.
Critically, timing matters enormously. Oral vitamin B3 given from day two of life did nothing — the pups still died. Injections starting at birth saved them. This suggests that NAXD disease demands early diagnosis and immediate intervention. The authors advocate for adding NAXD to neonatal screening panels, noting that circulating serine levels could serve as a practical biomarker to monitor whether treatment is working.
There are also tantalising human data points. Several published case reports describe NAXD patients receiving vitamin cocktails containing niacin, with apparent clinical benefit. These are anecdotal — small numbers, variable doses, no controls — but the direction matches the mouse data. The framework also nominated 14 other diseases as potentially B3-responsive, and a parallel B2 screen pointed to conditions including GPX4 deficiency (linked to ferroptosis) and the already-confirmed Brown-Vialetto-Van Laere syndrome. The nutrigenomics screening approach, in other words, isn't a one-off — it's a template.
The Questions That Still Need Answering
The study has real limitations the authors don't hide. The mouse model knocked out all NAXD isoforms simultaneously — but the human disease is more complex, with different isoforms operating in different cellular compartments. It's possible that some of the biology is compartment-specific in ways the model can't capture. The researchers also haven't established whether NAXD patients need lifelong vitamin B3 supplementation or whether treatment could eventually taper after a critical developmental window closes.
The dose translation from mouse to human is non-trivial. The 750 mg/kg injected daily in mice doesn't map directly to a clinical protocol — a proper dose-finding study in human patients will be essential, and the delivery method matters too. Intraperitoneal injection works in neonatal mice; it's not how you'd treat a newborn in a NICU. Oral formulations that achieve adequate brain NAD levels in a human infant are an open engineering problem.
And NADHX accumulation may matter beyond NAXD disease entirely. Recent studies have detected molecules with the same mass as NADHX building up in cardiac tissue after myocardial infarction — raising the possibility that NAXD inhibition or NADHX toxicity plays a role in common conditions like heart attack. That's speculative for now, but it's the kind of question this research makes urgent to ask.
- Vitamin B3 is already available — This isn't a drug waiting for trials; niacin and nicotinamide riboside are on pharmacy shelves, which could dramatically shorten the path to patient access if clinical data confirms the mouse results.
- Newborn screening is the bottleneck — The 48-hour treatment window means NAXD must be detected at birth, not after symptoms appear; adding it to neonatal panels may save lives immediately.
- The nutrigenomics template scales — The same CRISPR screening framework can be applied to all 13 classical vitamins and 50+ micronutrients, potentially nominating dozens more treatable rare diseases hiding in plain sight.
"These findings establish a nutritional genomics framework and demonstrate the therapeutic potential of precision vitamin interventions." — Garg, Blume, Huynh et al., Cell, 2026.
📄 Source & Citation
Primary Source: Garg A, Blume SY, Huynh H, et al. (2026). Vitamin B2 and B3 nutrigenomics reveals a therapy for NAXD disease. Cell, 189(12), 2648–2662. https://doi.org/10.1016/j.cell.2026.01.022 ↗
Authors & Affiliations: Ankur Garg & Skyler Y. Blume (co-first authors), Isha H. Jain (lead contact) — Gladstone Institutes & Department of Biochemistry and Biophysics, UCSF, San Francisco, CA; Arc Institute, Palo Alto, CA. Additional collaborators from Cornell University, University of Chicago, UT Health San Antonio, and University of Florida.
Data & Code: snRNA-seq dataset available at GEO: GSE304377 ↗. Analysis code: github.com/gladstoneinstitutes/GB-SB-1467 ↗
Key Themes: Nutrigenomics · NAD Metabolism · Rare Disease · CRISPR Screening · Neonatal Therapy
Supporting References:
[1] Van Bergen NJ et al. (2019). NAD(P)HX dehydratase (NAXD) deficiency: a novel neurodegenerative disorder exacerbated by febrile illnesses. Brain, 142(1):50–58. doi.org/10.1093/brain/awy310 ↗
[2] Manor J et al. (2022). Niacin therapy improves outcome and normalises metabolic abnormalities in an NAXD-deficient patient. Brain, 145:e36–e40. doi.org/10.1093/brain/awac065 ↗
[3] Walvekar AS et al. (2025). Failure to repair damaged NAD(P)H blocks de novo serine synthesis in human cells. Cell Mol Biol Lett, 30:3. doi.org/10.1186/s11658-024-00681-8 ↗
No comments yet. Be the first to share your thoughts.
Leave a Comment