In This Article
- The Discovery That Changed How We See GLP-1 Drugs
- Why the Old Story Was Only Half Right
- How Do GLP-1 Drugs Actually Quiet Food Cravings in the Brain?
- What This Means Beyond the Weight-Loss Clinic
- The Questions That Still Need Answering
Picture the moment you open a bag of crisps "just for one" and somehow reach the bottom. That isn't a failure of willpower — it's your brain's reward system doing exactly what it was designed to do, flooding your nucleus accumbens with dopamine the second palatable food arrives. Now, scientists at the University of Virginia have discovered that GLP-1 receptor agonists — the blockbuster drug family behind Ozempic, Wegovy, and a new generation of daily pills — can quietly turn that signal down, through a brain circuit nobody was looking at.
The Discovery That Changed How We See GLP-1 Drugs
Published in Nature in May 2026, the study set out to understand how newer oral GLP-1 drugs suppress appetite — not just that they do. The research, led by Elizabeth Godschall, Taha Bugra Gungul, and Isabelle Sajonia, tackled a problem that had been quietly frustrating the field for years.
Most GLP-1 drugs on the market — semaglutide (Ozempic/Wegovy), liraglutide — are injections with complex manufacturing chains. Two newer, small-molecule versions, danuglipron (Pfizer) and orforglipron (Eli Lilly), can be swallowed as a daily pill. That's a game-changer for accessibility. But there was a catch: these small molecules bind to the human version of the GLP-1 receptor, not the mouse version, making it nearly impossible to study their brain effects in standard lab animals.
The team solved this with elegant genetic engineering — swapping a single amino acid in the mouse's GLP-1 receptor gene to create a "humanised" mouse. The result was a model that responds to the same pill-form drugs humans take, opening the door to the neurological investigation that followed.
Why the Old Story Was Only Half Right
Before this study, the accepted explanation for how GLP-1 receptor agonists reduce eating went roughly like this: the drugs activate receptors in the hypothalamus and brainstem, regions that handle basic hunger and satiety. Think of these as the body's fuel gauge — they tell you when the tank is full. That part is true. But it only explains why people eat less at mealtimes.
What it doesn't explain is why many patients on these drugs also stop reaching for chocolate at 11 pm when they're not even hungry. That's hedonic eating — eating for pleasure and reward, not fuel. The neuroscience of hedonic feeding runs through the brain's dopamine-powered reward system, the same circuitry involved in addiction. And until now, nobody had pinned down exactly how GLP-1 drugs touch it.
How Do GLP-1 Drugs Actually Quiet Food Cravings in the Brain?
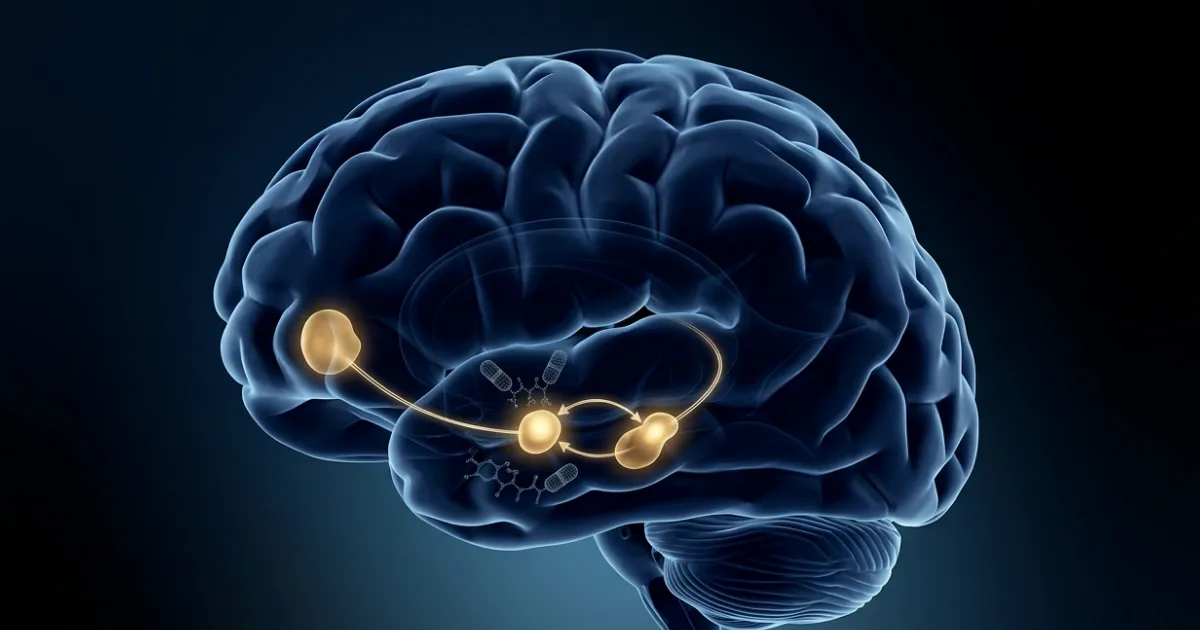
The answer lies in a walnut-sized structure deep in the brain called the central amygdala — better known for processing fear and emotion than appetite. The researchers found a small, previously overlooked cluster of GLP-1 receptor-expressing neurons sitting there, and when the drugs activated them, something remarkable happened: the mice drastically reduced their consumption of high-fat, palatable food, while leaving their normal meals virtually untouched.
To confirm this wasn't coincidence, the team used fibre-optic probes to watch dopamine levels in real time as mice ate. After a dose of danuglipron or orforglipron, the usual dopamine spike in the nucleus accumbens — the brain's pleasure hub — was dramatically blunted the moment a mouse reached for high-fat food. The reward signal that makes junk food feel compulsive was quietened, not abolished, but measurably quietened.
The circuit they traced runs like this: GLP-1 drugs activate inhibitory (GABA-releasing) neurons in the central amygdala → those neurons send projections to the ventral tegmental area (VTA), the brain's dopamine factory → the VTA then releases less dopamine into the nucleus accumbens. It's a three-stop relay that puts a dimmer switch on the brain's reward response to pleasurable food. Activating this amygdala circuit with light pulses alone (using optogenetics) reproduced the same selective reduction in hedonic eating — proving the circuit is both necessary and sufficient.
"These findings identify a neural circuit through which small-molecule GLP-1 receptor agonists modulate reward processing, with implications for the treatment of substance-use disorder and binge eating."
— Godschall, Gungul, Sajonia et al., University of Virginia · Nature, 2026What This Means Beyond the Weight-Loss Clinic
If a drug can silence the dopamine response to a chocolate bar, could it do the same for alcohol, opioids, or nicotine? The question is no longer just theoretical. Real-world data from Nature Communications already show that patients on semaglutide have significantly lower rates of alcohol use disorder relapse. A separate analysis in Molecular Psychiatry found similar patterns for cannabis use.
This new study provides the first concrete circuit-level explanation for why. The central amygdala sits at an anatomical crossroads between the brain's emotional processing system and its dopamine reward highway — precisely the junction where addiction takes hold. Knowing that GLP-1 receptor agonists engage this junction directly gives researchers a specific target to design more precise therapies for binge-eating disorder and compulsive substance use.
There is also a practical dimension for the hundreds of millions of people who may one day take these drugs. Orforglipron was approved by the FDA for weight management earlier this year. Unlike injections, an oral pill is manufacturable at scale and far cheaper — Eli Lilly's CEO has called it "a product for the masses." Understanding exactly how it acts on the brain is not just academic; it shapes dosing, safety monitoring, and the conditions it might eventually treat.
The Questions That Still Need Answering
Compelling as the findings are, the researchers are clear about what they don't yet know. These experiments were conducted in mice — and while the humanised receptor model is a major advance, the human brain's amygdala is vastly more complex. Whether the same three-node circuit operates identically in people taking these drugs remains to be confirmed with brain-imaging studies in humans.
There are also open questions about long-term effects. The dopamine system is extraordinarily adaptable — it recalibrates when chronically dampened, which is part of why addiction is so hard to treat. Whether years of GLP-1-mediated suppression of reward signalling might alter motivation, mood, or the experience of pleasure in other domains is genuinely unknown. Researchers writing in Science have already flagged the need for vigilance as these drugs reach wider populations. The amygdala circuit the Virginia team identified is not just a therapeutic target — it is a reason to watch carefully.
- A new brain map for obesity drugs — The central amygdala→VTA→NAc circuit is now a confirmed target for GLP-1 action, not a hypothesis. Future drugs can aim for it directly.
- Hedonic eating has its own switch — This study shows that craving-driven eating and hunger-driven eating are biologically distinct, controlled by parallel but separate circuits. That changes how clinicians should think about treating binge-eating disorder.
- Addiction medicine may be transformed — If GLP-1 pill formulations can dampen the dopamine response to both food and drugs of abuse, the same mechanism that trims waistlines could one day help treat alcohol or opioid dependence.
"As small-molecule compounds such as orforglipron move swiftly towards widespread clinical use, it will be crucial to define their long-term effects on mesolimbic circuitry and motivated behaviours." — Godschall, Gungul, Sajonia et al., Nature, 2026.
📄 Source & Citation
Primary Source: Godschall EN, Gungul TB, Sajonia IR, et al. (2026). A brain reward circuit inhibited by next-generation weight-loss drugs in mice. Nature. https://doi.org/10.1038/s41586-026-10444-4
Authors & Affiliations: Elizabeth N. Godschall, Taha Bugra Gungul, Isabelle R. Sajonia, and colleagues — Department of Biology & Neuroscience Graduate Program, University of Virginia; University of California, Irvine; University of Washington.
Data & Code: All data available on LabArchives; code available at github.com/UVACircMetNeuLab/glp1r-reward-circuit
Key Themes: GLP-1 Receptor Agonists · Central Amygdala · Dopamine Reward · Hedonic Feeding · Addiction Neuroscience
Supporting References:
[1] Wharton S et al. (2023). Daily oral GLP-1 receptor agonist orforglipron for adults with obesity. N. Engl. J. Med., 389:877–888.
[2] Huang K-P et al. (2024). Dissociable hindbrain GLP1R circuits for satiety and aversion. Nature, 632:585–593.
[3] Wang W et al. (2024). Associations of semaglutide with incidence and recurrence of alcohol use disorder in real-world populations. Nat. Commun., 15:4548.
[4] Drucker DJ. (2024). The benefits of GLP-1 drugs beyond obesity. Science, 385:258–260.
[5] Alhadeff AL, Rupprecht LE & Hayes MR. (2012). GLP-1 neurons in the nucleus of the solitary tract project directly to the ventral tegmental area and nucleus accumbens to control for food intake. Endocrinology, 153:647–658.
No comments yet. Be the first to share your thoughts.
Leave a Comment