In This Article
- The Silent Epidemic Filling India's Clinics
- The Molecular Culprit Nobody Was Looking At
- Why Does miR-93 Cause So Much Damage?
- What This Means Beyond the Lab
- What Comes Next — and What's Still Unknown
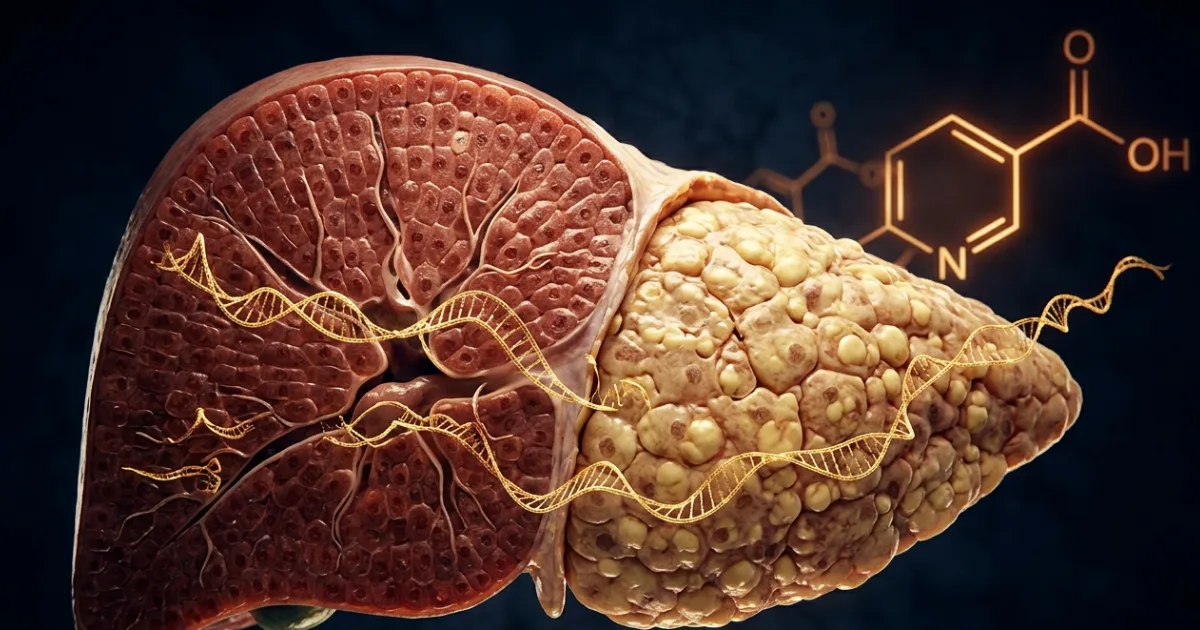
One in four people on Earth is walking around with a liver quietly drowning in fat — and most have no idea. Fatty liver disease, now formally called metabolic dysfunction-associated steatotic liver disease (MASLD), has become the world's most common chronic liver condition, outpacing alcohol-related liver damage and viral hepatitis combined. And yet, there is still not a single FDA-approved drug to treat it. A new study from researchers at UNIST, Pusan National University, and Ulsan University Hospital may have just changed the entire conversation — by identifying a single molecular switch at the heart of the problem, and a surprisingly familiar compound that can flip it back.
The Silent Epidemic Filling India's Clinics
Think of your liver as the body's most overworked factory — processing everything you eat, filtering toxins, packaging fats for transport, and keeping blood sugar in check. It performs over 500 distinct functions. Now imagine someone secretly jamming one of its core machines. That's essentially what's happening in MASLD.
Globally, MASLD affects an estimated 25–30% of the world's population. In India, where high-carbohydrate diets, sedentary lifestyles, and a genetic predisposition to abdominal fat storage collide, the numbers are alarming — some estimates suggest prevalence as high as 32% among urban adults. Left untreated, MASLD can progress through a gauntlet of worsening stages: from simple fat accumulation to liver inflammation (MASH), scarring (fibrosis), cirrhosis, and eventually liver cancer.
The problem is not just that fat builds up. It's why the liver's own fat-burning systems fail to contain it.
The Molecular Culprit Nobody Was Looking At
The researchers didn't set out hunting for a single villain. They began with a sweeping analysis of gene expression data from MASLD patients and healthy controls — essentially asking the question: which genes are quieted in a diseased liver, and what might be silencing them?
The answer pointed to a family of tiny molecules called microRNAs. These are not genes themselves, but short snippets of RNA that act like volume knobs — turning down the production of specific proteins. Among hundreds of candidates, one stood out: miR-93, a member of the miR-17 family, was dramatically elevated in the liver tissue of MASLD patients. The more severe the disease — measured by liver enzyme levels, BMI, and a standardized liver damage score — the higher the miR-93 levels.
The team confirmed this across multiple independent datasets from both human patients and mouse models. They even found the same pattern in liver cells exposed to fatty acids in a dish. Something about fat overload was turning up miR-93 — and miR-93, in turn, was making things worse.
Why Does miR-93 Cause So Much Damage?
Here's where the science gets genuinely elegant. miR-93 works by suppressing a protein called SIRT1 — often called the "longevity protein" because of its role in energy balance, metabolism, and cellular repair. SIRT1 is essentially a master coordinator: it activates a chain of proteins (LKB1 → AMPK) that tells liver cells to burn fat and pump the brakes on producing new cholesterol. When SIRT1 is working properly, the liver is a lean, efficient machine.
When miR-93 levels are high, SIRT1 gets muzzled. The LKB1-AMPK signalling cascade shuts down. Fat burning (fatty acid oxidation) slows. Cholesterol and fat synthesis ramp up. Mitochondria — the cell's power generators — become sluggish. The liver starts hoarding fat instead of burning it, and the resulting buildup triggers inflammation and, over time, scarring.
To prove this wasn't coincidence, the researchers did something clever: they bred mice that completely lack the miR-93 gene. Fed a high-fat, high-fructose diet designed to trigger MASLD, these knockout mice stayed significantly leaner than normal mice. Their livers showed less fat, less inflammation, less fibrosis. Their blood sugar was better controlled. Their mitochondria were more active. Then the team reversed the experiment — injecting extra miR-93 into normal mice — and watched the exact opposite happen: the disease accelerated.
"miR-93 deficiency mitigates hepatic steatosis and metabolic dysfunction, whereas miR-93 overexpression exacerbates these pathologies."
— Choi JH et al., UNIST · Research Square, 2024The effect held up across multiple mouse models — including a genetic obesity model and a diet specifically designed to mimic advanced human liver disease. Crucially, when the team knocked out SIRT1 even in the miR-93-deficient mice, the protective effect disappeared. That was the smoking gun: the damage from miR-93 runs directly through SIRT1.
What This Means Beyond the Lab
The most immediately exciting part of this research isn't the molecular mechanism — it's what the team found when they went hunting for a drug to exploit it. Using a high-throughput screen of 150 FDA-approved compounds, they built a system where a "reporter" gene would glow brighter whenever a drug blocked miR-93 from being produced. The top hit? Niacin — also known as vitamin B3, and already stocked in pharmacies from Mumbai to Manchester.
Niacin is not new. It's been used for decades to lower blood triglycerides and raise HDL ("good") cholesterol. But this study reveals a mechanism nobody had formally connected before: niacin suppresses miR-93 expression in the liver, which allows SIRT1 to do its job, which in turn restores normal fat metabolism. When mice with MASLD were given niacin supplements alongside their high-fat diet, their liver fat dropped, inflammation decreased, and key metabolic markers improved — but only in mice with normal miR-93 levels. When researchers artificially forced miR-93 to stay high, niacin's benefits evaporated. That's a critical finding: it suggests that patients with very high miR-93 levels may not respond to niacin alone, and could need a direct miR-93 inhibitor instead.
What Comes Next — and What's Still Unknown
This research is a preprint — it has not yet completed peer review, so it should be read as promising evidence rather than settled fact. The mouse models, while carefully chosen, do not perfectly replicate human liver disease. The study also does not tell us the optimal niacin dose for humans targeting the miR-93 pathway, or whether long-term niacin use in MASLD patients is safe given known side effects like flushing and, at high doses, potential liver stress of its own. The researchers acknowledge that patients with elevated miR-93 may need direct miR-93 inhibitors — a class of drugs that exist in early clinical development for other conditions but have not yet been tested in MASLD.
What the field needs next is a human clinical trial measuring miR-93 levels in MASLD patients, testing niacin's effect on those levels, and tracking liver outcomes. That work would likely take years. But the molecular map drawn here — miR-93 suppresses SIRT1, which breaks the LKB1-AMPK brake on fat synthesis — gives researchers a clear target for that next chapter.
- miR-93 is a gatekeeper — Elevated levels of this microRNA actively drive fat accumulation, inflammation, and fibrosis in the liver by silencing SIRT1, making it a compelling target for MASLD therapy.
- Niacin has a new trick — Beyond its established role in cholesterol management, vitamin B3 suppresses miR-93 expression, suggesting it could benefit MASLD patients with normal or moderate miR-93 levels via a newly understood mechanism.
- Personalised treatment may be key — The niacin benefit disappears when miR-93 is forced high, hinting that future MASLD treatment could depend on measuring a patient's miR-93 levels before choosing a therapy.
"The modulation of the miR-93/SIRT1 axis, particularly through niacin treatment, presents a potential therapeutic avenue for MASLD, a disease with few current treatment options." — Choi JH et al., Research Square, 2024.
📄 Source & Citation
Primary Source: Choi JH, Lee YH, Lee J, Jeong J, Park K, et al. (2024). Hepatic miR-93 promotes the pathogenesis of metabolic dysfunction-associated steatotic liver disease by suppressing SIRT1. Research Square (preprint). https://doi.org/10.21203/rs.3.rs-5202645/v1
Authors & Affiliations: Jang Hyun Choi (UNIST, lead corresponding author), Hwayoung Yun (Pusan National University), Neung Hwa Park (Ulsan University Hospital), and collaborators across KRIBB, GIST, and Sungkyunkwan University.
Data & Code: RNA sequencing data deposited in NCBI Gene Expression Omnibus under accession code GSE277560.
Key Themes: MASLD · MicroRNA · SIRT1 · Lipid Metabolism · Niacin Therapy
Supporting References:
[1] Younossi ZM et al. (2023). The global epidemiology of nonalcoholic fatty liver disease (NAFLD) and nonalcoholic steatohepatitis (NASH): a systematic review. Hepatology, 77(4):1335–1347. PubMed
[2] Hou XY et al. (2008). SIRT1 regulates hepatocyte lipid metabolism through activating AMP-activated protein kinase. Journal of Biological Chemistry, 283(29):20015–20026. PubMed
[3] Gjorgjieva M et al. (2019). miRNAs and NAFLD: from pathophysiology to therapy. Gut, 68(11):2065–2079. PubMed
No comments yet. Be the first to share your thoughts.
Leave a Comment